 GET SAVINGS CARD
GET SAVINGS CARDZIPSOR® (diclofenac potassium) is a prescription Non-Steroidal Anti-Inflammatory Drug (NSAID). ZIPSOR is used for relief of mild-to-moderate pain in adults (18 years of age and older).
About ZIPSOR, a non-opioid approach
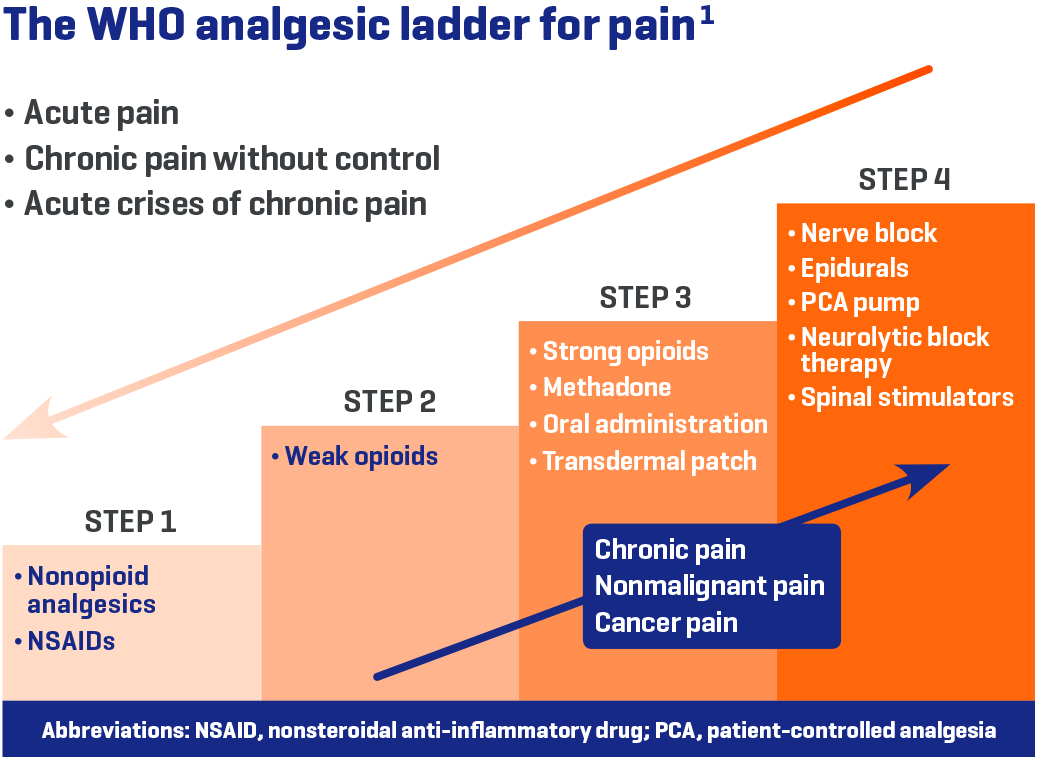
World Health Organization (WHO) recommendations for pain relief1
- Start with non-opioid analgesics, including nonsteroidal anti-inflammatory drugs (NSAIDs)
- Usage (with or without adjuvants) is suggested at every step in care
The US government’s Centers for Disease Control and Prevention (CDC) recommends that2:
- Acute pain may be managed without opioids
- Non-opioid options include NSAIDs
- Discuss with your patients about ways to relieve pain that do not involve prescription opioids
NSAIDs have been used for pain for nearly 60 years and are widely prescribed and recommended by physicians.
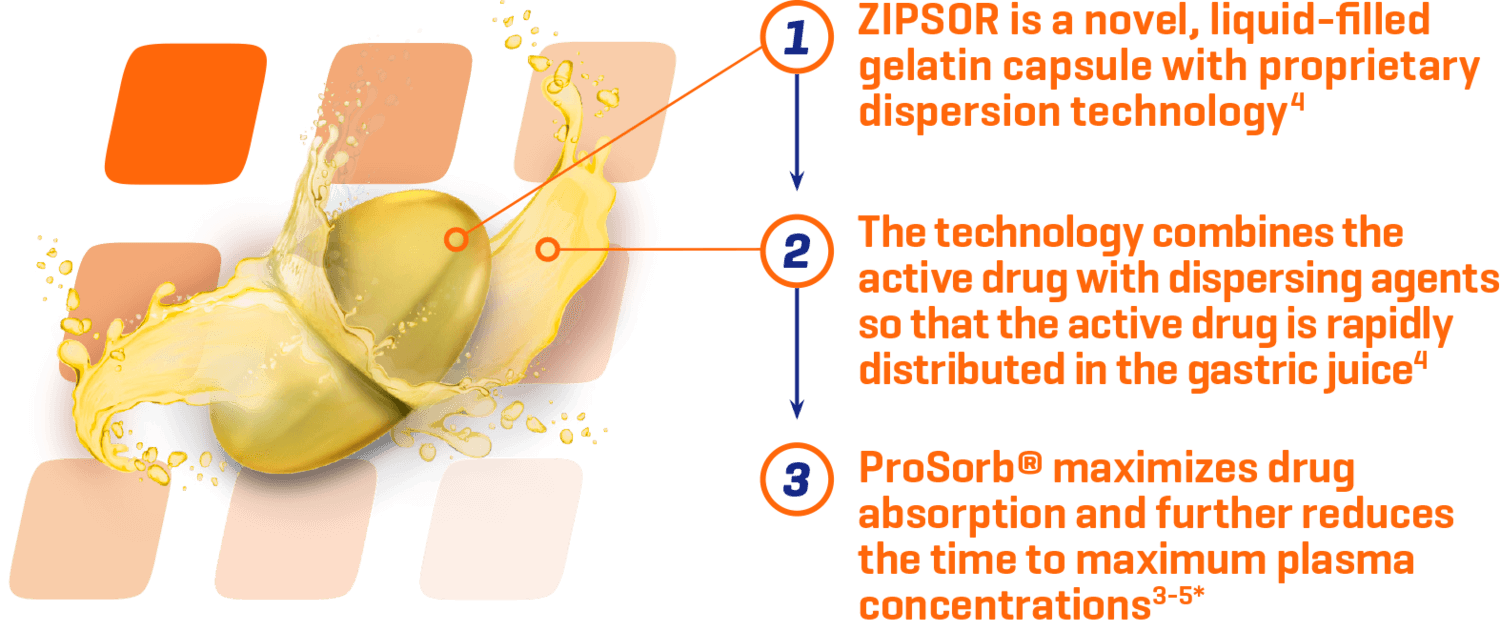
Why ZIPSOR?
ProSorb® technology maximizes absorption3
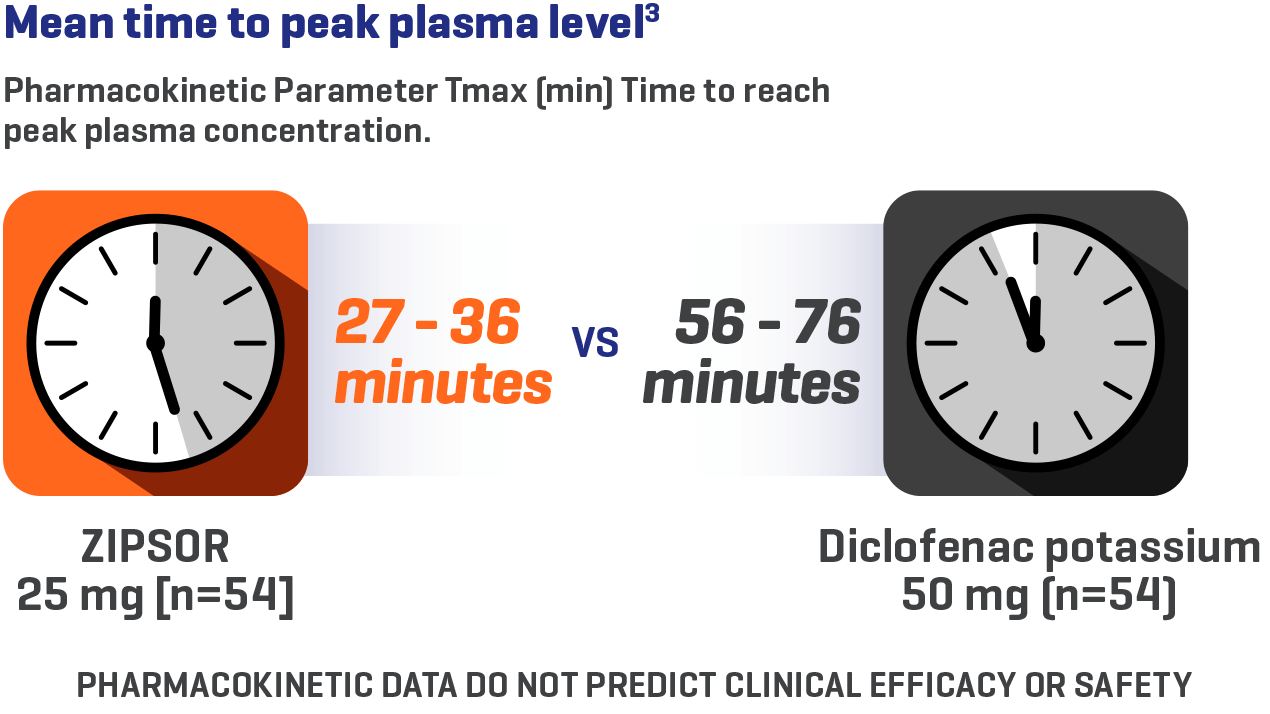
ZIPSOR is designed for rapid, consistent absorption3*
*Study conducted in healthy volunteers ≥ 18 years of age.
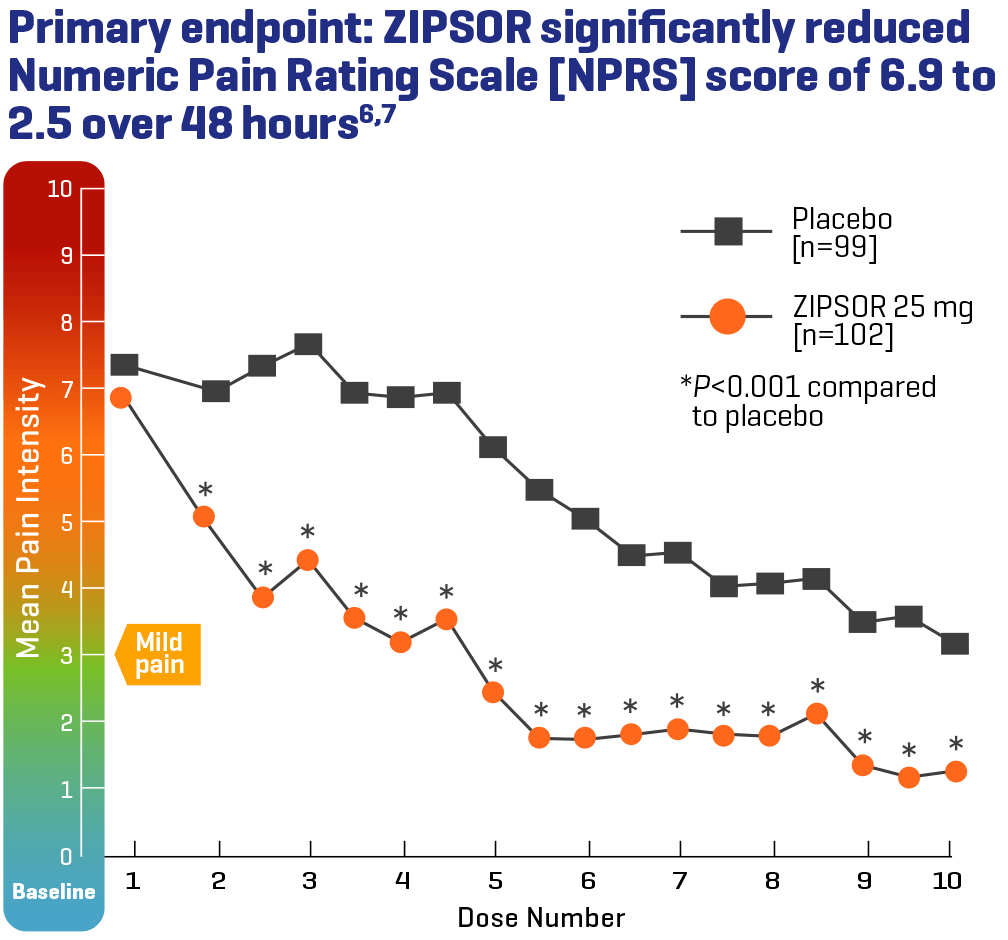
Significant pain reduction with every dose6,7
Primary endpoint: pain reduction over 48 hours after bunionectomy6,7
Bunionectomy surgery results in considerable pain and is a standard model for assessing efficacy of an acute pain medication.8
Study Design: This Phase III, randomized, double-blind, parallel-group, placebo-controlled study was conducted in a single-dose period and a multiple-dose period. Patients with a qualifying level of postsurgical pain (score ≥4 on an 11-point NPRS from 0=no pain to 10=worst possible pain) were randomly assigned to receive ZIPSOR 25 mg or placebo soft gelatin capsules in a 1:1 ratio.7
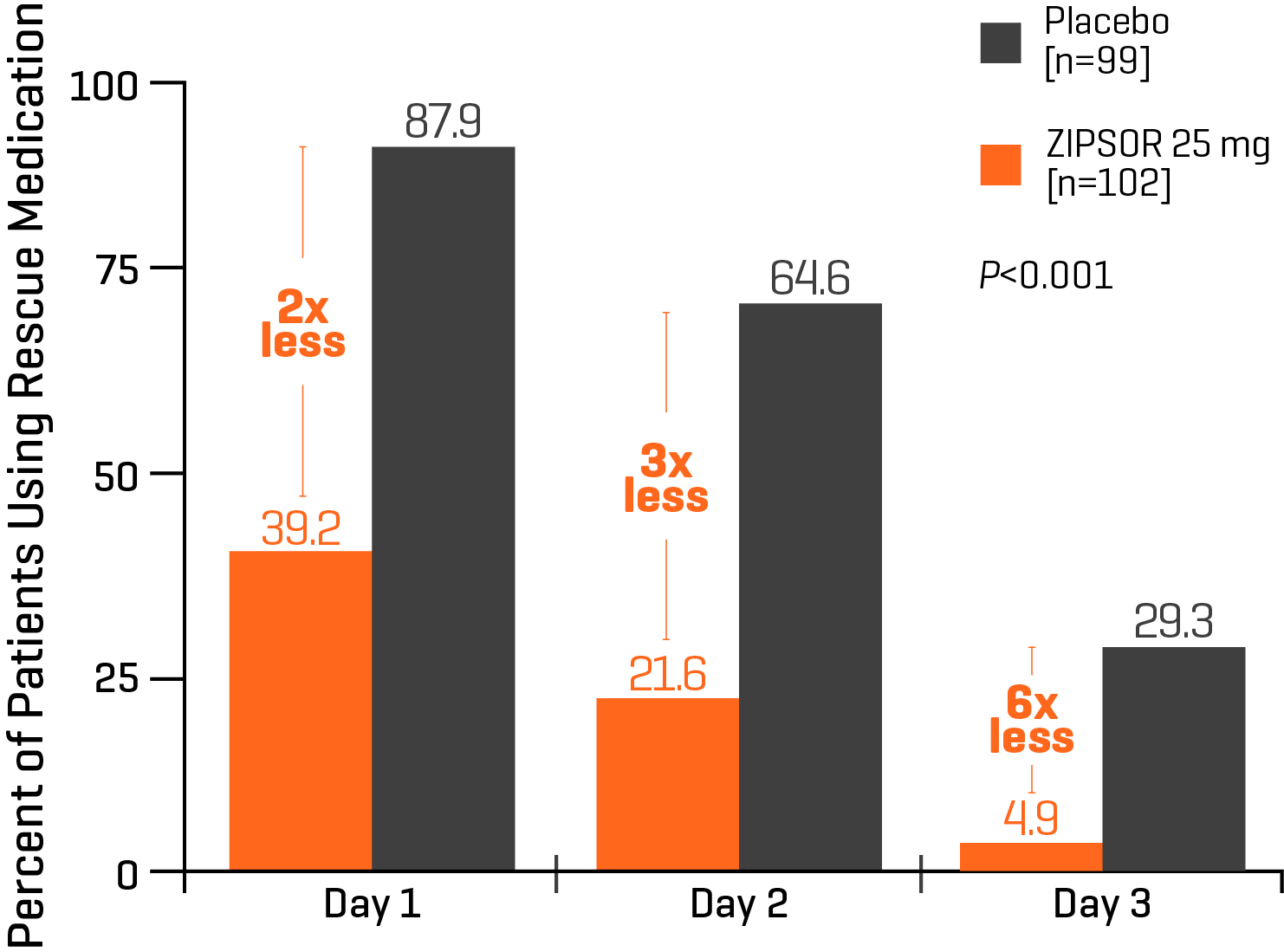
Use of rescue medication (1-2 hydrocodone/acetaminophen tablets) during the inpatient 48-hour multiple-dose period after bunionectomy in patients who received ZIPSOR 25 mg or placebo. Day 3 was not a full day, and the data reflect use of rescue medication only up to the time of discharge.7
Your patients can save on ZIPSOR
Your patients can save money on ZIPSOR by downloading a copay card.*
*Terms and conditions may apply. View savings card for full terms and conditions.
Please see Important Safety Information and full Prescribing Information, including BOXED WARNING.